
We are Pinco Biotherapeutics
We are on a mission to develop precise, durable, and non-addictive therapies for chronic neuropathic pain.
Neuropathic pain arises from dysfunction of the nervous system and affects patients with Diabetic Peripheral Neuropathy, Chemotherapy-Induced Peripheral Neuropathy, and Small Fiber Neuropathy.
Despite the scale of the problem, treatment options remain limited. First-line therapies (e.g., gabapentinoids, SNRIs) offer modest efficacy. Later-line options carry serious safety and addiction concerns.
This often leaves patients having to choose between pain, or addiction. This is a choice we don’t think anyone should have to make.
We believe pain treatment should be mechanistically targeted, durable, non-addictive, cost-conscious to manufacture, and expandable across multiple pain indications.
PincoBio was founded to bring molecular precision to a therapeutic area long dominated by systemic drugs.


Hear from our CEO
Our Technology
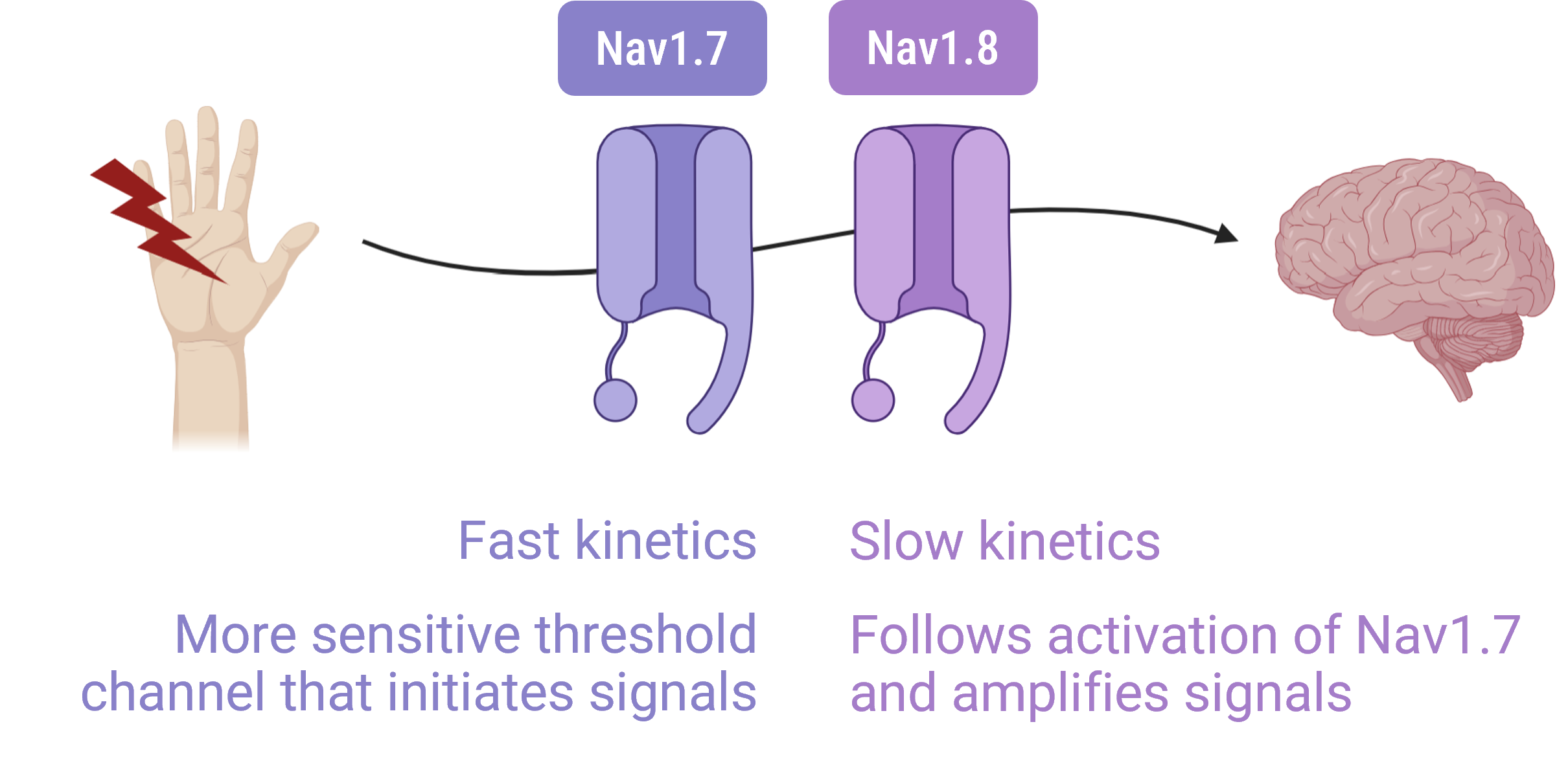
Pathological pain is driven by specific electrical signals in our neurons, controlled by tiny gates called sodium channels. Two of these are critical: Nav1.7 and Nav1.8. While they have differing kinetics are roles in pain signaling, much of the recent focus in pain has been on Nav1.8.

We’re choosing to focus on Nav1.7. We’ve chosen this because it is highly expressed in dorsal root ganglion (DRG) neurons and has strong genetic validation:
Gain-of-function mutations lead to severe pain syndromes
Loss-of-function mutations lead to congenital insensitivity to pain
We believe Nav1.7 is the master switch for pain. By turning off this specific switch, we can stop pain at its source.
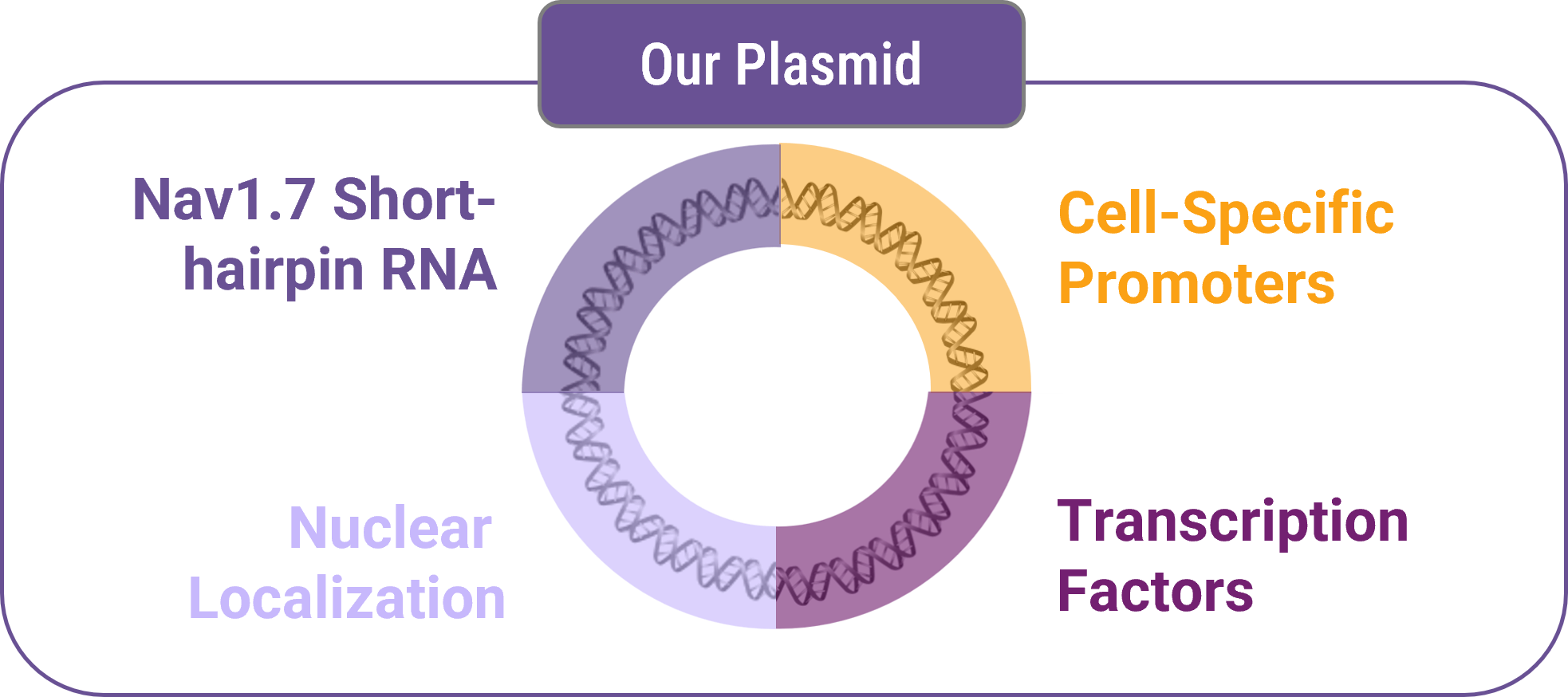
Despite strong validation, small molecule inhibitors have struggled with specificity and off-target effects. So we’re innovating our approach and developing a non-viral DNA-based Plasmid.
Our plasmid contains:
Nav1.7-targeting shRNA
Cell-specific promoters
Nuclear localization sequences
Transcription factor optimization elements
This design allows targeted suppression of SCN9A (Nav1.7 gene) expression.
Our Timeline

We are completing our proof of concept studies in DRG neurons and are moving into plasmid optimization and animal studies.
We anticipate signing a licensing deal for our therapeutic asset based on Phase I results in 2030
Contact us
We're always looking to connect with investors, collaborators, or other partners!
Send us a message!
